Malaria Vaccine Rts S As01
RTSSAS01 is a preerythrocyticPlasmodium falciparum malaria candidate vaccine. The World Health Organization on Wednesday endorsed the RTSSAS01 malaria vaccine the first against the mosquito-borne disease that kills more than 400000 people a year mostly African children.
First Results of Phase 3 Trial of RTSSAS01 Malaria Vaccine in African Children.

Malaria vaccine rts s as01. This two-vial presentation was the chosen. This recommendation is based on results from an ongoing pilot program in Ghana Kenya and Malawi that has. Malaria vaccine RTSSAS02A in malaria-naive adults.
N Engl J Med2011 Oct 18. Article Google Scholar 6. Pilot vaccine implementation has recently begun in 3 African countries.
Over 800000 children have received the vaccine so far and have been benefiting from the additional protection against the disease. VE was higher in children than in infants but even at modest levels of VE the number of malaria. More than a dozen vaccine candidates are now in clinical development and one GlaxoSmithKline Biologicals RTSS has completed Phase III clinical testingthe first malaria vaccine candidate to advance this far.
The World Health Organization on Wednesday endorsed the RTSSAS01 malaria vaccine the first against the mosquito-borne disease that kills more than 400000 people a. The RTSS vaccine has been approved as it significantly reduces malaria and life-threatening severe cases. RTSSAS01 is a malaria vaccine candidate that has undergone phase 3 evaluation across several sites in Africa that have varying intensities of malaria.
As this concerning trend calls for new tools to combat the disease the RTSS vaccine has arrived just in time. The malaria vaccine candidate RTSSAS01 is presented as a two-vial formulation with the antigen in lyophilized form and the liposome-based Adjuvant System AS01 in liquid form. The malaria vaccine candidate RTSSAS01 also known as Mosquirix consists of hepatitis B surface antigen virus-like particles incorporating a portion of the Plasmodium falciparum-derived circumsporozoite protein and a liposome-based adjuvantThe clinical development of the malaria vaccine RTSSAS01 reached a critical stage in 2015 with the publication of the results of a large.
The candidate malaria vaccine RTSSAS01 reduced episodes of both clinical and severe malaria in children 5 to 17 months of age by approximately 50 in. Of a sustainable malaria control program and an important tool for the malaria elimination technical roadmap 4. RTSS Clinical Trial Partnerships Efficacy and safety of RTSSAS01 malaria vaccine with or without a booster dose in infants and children in Africa.
RTSS Clinical Trials Partnership. On 6 October 2021 WHO recommended widespread use of the RTSS malaria vaccine among children. The lyophilized antigen needs to be reconstituted using the liquid AS01 adjuvant prior to administration of the vaccine.
RTSSAS01 prevented many cases of clinical and severe malaria over the 18 mo after vaccine dose 3 with the highest impact in areas with the greatest malaria incidence. The World Health Organization WHO is recommending widespread use of the RTSSAS01 RTSS malaria vaccine among children in sub-Saharan Africa and in other regions with moderate to high P. Final results of a phase 3 individually randomised controlled trial.
The decision followed a review of a pilot programme deployed since 2019 in Ghana Kenya and Malawi where more than two million doses were given of the vaccine first made by. The vaccine was created in 1987 and began pilot implementation in endemic countries in 2019. The recommendation is based on results from an ongoing pilot programme in Ghana Kenya and Malawi that has reached more than 800 000 children.
The RTS S AS01 malaria vaccine should be given on a 4-dose schedule to children from the age of 5 months to reduce disease and the burden of malaria the WHO prescribes. Indeed in children aged 5 to 17 months who received four doses of RTS S AS01 the vaccine prevented about four in ten cases of malaria during 4 years of follow-up. To optimize vaccine implementation visits for young children it could be efficient to administer the first RTSSAS01 malaria vaccine dose during the Expanded Programme on Immunization EPI visit at 6 months of age together with Vitamin A supplementation and the third RTSSAS01 dose on the same day as yellow fever YF measles and rubella vaccines at 9 months of.
The RTSSAS01 vaccine against Plasmodium falciparum malaria infection completed phase III trials in 2014 and demonstrated efficacy against clinical malaria of approximately 36 over 4 years for a 4-dose schedule in children aged 517 months. The vaccine significantly reduces malaria and life-threatening severe malaria in young African children. Malaria Consortium - Who recommends rts s as01 malaria vaccine for children in sub saharan africa - One of the worlds leading non-profit organisations specialising in the prevention control and treatment of malaria and other communicable diseases among vulnerable populations.
In October 2021 the vaccine was endorsed by the World Health Organization WHO for broad use in children making it the first malaria vaccine candidate and first vaccine to address parasitic infection to receive this recommendation. RTSSAS01 trade name Mosquirix is a recombinant protein -based malaria vaccine. Efficacy and safety of RTSSAS01 malaria vaccine with or without a booster dose in infants and children in Africa.
Is recommending the broad use of the worlds first malaria vaccine. The vaccine is a self-assembling virus-like particle vaccine containing a fusion protein RTS of the NANP repeat and C-terminal portions R and T respec-. WRAIR research on RTSSAS02 25-26 continues to be a vital part of malaria.
RTSSAS01 RTSS is a vaccine that acts against Plasmodium falciparum the deadliest malaria parasite globally and the most prevalent in Africa. Final results of a phase 3 individually randomised controlled trial. World Health Organization Malaria vaccine technology roadmap.
But reductions in numbers of cases have stalled over the last few years and incidence may have increased.
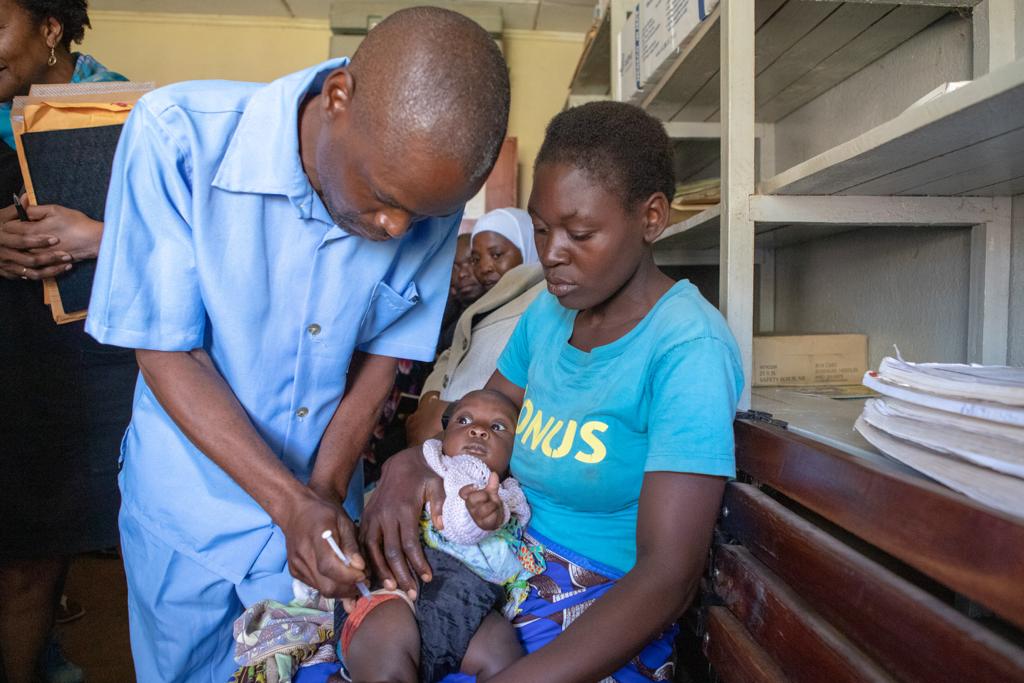
World S First Malaria Vaccine Rts S As01 Mosquirix

Jci Advances And Challenges In Malaria Vaccine Development

Proposed Immunological Mechanism Determining Protection Or Lack Download Scientific Diagram
Malaria Vaccines Recent Advances And New Horizons

Systems Analysis Of Protective Immune Responses To Rts S Malaria Vaccination In Humans Pnas

The Rts S As Malaria Vaccine Candidate A Status Review Semantic Scholar
Malaria Vaccine In Young African Children Youtube

A Structure Of The Rts S Antigen Schematic Description Of The Download Scientific Diagram
Rts S As01 Rts S Mosquirix New Drug Approvals

Ammren On Twitter A First Generation Vaccine Known As Rts S As01 Rts S Has The Potential To Strengthen Efforts To Control Malaria In Africa And Save Tens Of Thousands More Young Lives Ammren Malaria Accra Ghana

Immunogenicity Of The Rts S As01 Malaria Vaccine And Implications For Duration Of Vaccine Efficacy Secondary Analysis Of Data From A Phase 3 Randomised Controlled Trial The Lancet Infectious Diseases


0 Response to "Malaria Vaccine Rts S As01"
Post a Comment